 Electrochemical studies reveal that (phen 2N 2)Fe has a relatively high Fe(III/II) potential with a correlated ORR onset potential within 150 mV of Fe-N-C. N 1s XPS and XAS signatures for (phen 2N 2)Fe are remarkably similar to those of Fe-N-C. Herein, we report a molecular pyridinic hexaazacyclophane macrocycle, (phen 2N 2)Fe, and compare its spectroscopic, electrochemical, and catalytic properties for ORR to a typical Fe-N-C material and prototypical pyrrolic iron macrocycles. A leading postulate is that the iron-containing active sites exist primarily in a pyridinic Fe-N 4 ligation environment, yet, molecular model catalysts generally feature pyrrolic coordination. 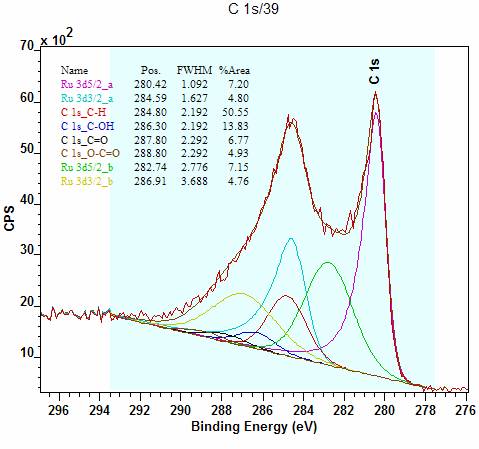
Iron- and nitrogen-doped carbon (Fe-N-C) materials are leading candidates to replace platinum catalysts for the oxygen reduction reaction (ORR) in fuel cells however, their active site structures remain poorly understood. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
